In a notice sent to doctors and seen by TheCable, the company said the decision to recall was reached after some counterfeit products were found within the legitimate supply chain.
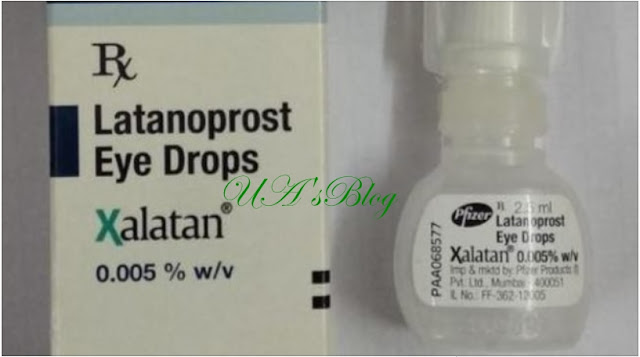
“Please be advised that Pfizer is recalling, at the patient level, the below lots of Xalatan 0.005%w/v Eye drops solution (the “Product Lots”) from distribution in Nigeria,” it said.
“The cause of this voluntary recall is due to the identification of two separate confirmed counterfeit Xalatan 0.005% w/v Eye drops 2.5ml solution lots, with authentic Pfizer Lot numbers W6769 and AK4753, being distributed in the legitimate supply chain in the Nigeria market where the authentic Xalatan 0.005% w/v Eye drops 2.5ml solution with the same lot numbers been distributed.
READ ALSO: Presidential petition panel: Atiku expresses worry over delay in naming justices
“The use of the counterfeit Product Lots has a high probability to cause therapeutic failure and adverse events, such as infections. The potential risk to patients is considered high as patients will not be able to identify the counterfeit product.”
The product batches affected are those with lot numbers W67369; expiring 10/2020 and AK4753 expiring 10/2021
Pfizer said the recall plan is to immediately stop distribution and place the product lots into quarantine.
The company said the recall is being carried out with the knowledge and approval of the National Food and Drug Administration and Control Agency (NAFDAC).
TheCable has contacted Abubakar Jimoh, the agency’s spokesperson, for comments.
READ ALSO: Kano 9 abduction is criminal NOT ethnic, says Onitsha traditional council
Are you an artiste? Do you want your music to go viral and reach a large number of audience? Promote your music and Submit your story on Ujuayalogusblog.com by clicking here. For Advert Inquiries Tel/+44(0)7590363984
For More: Subscribe to Ujuayalogusblog.com










No comments:
Post a Comment